|
Introduction
Systemic lupus Erythematosus
(SLE) is an autoimmune disease with wide clinical features.
The diagnosis of SLE is based on American College of
Rheumatology (ACR) and SLICC criteria. SLE affects usually
young adults. Few reports studied SLE in the elderly
[1,2,3,4].
We enrolled a retrospective study to establish the epidemiological,
clinical and laboratory profile, therapeutic and prognostic
aspects and course disease of late onset SLE. The aim
of our study is to compare the different presentations
of SLE in old and young population.
Patients
and Method
We conducted a cross-sectional study that consisted
of 83 SLE patients (75 female and 8 male), who attended
the Department of Internal Medicine in Monastir University
Hospital between 2006 and 2015. All patients met at
least 4 criteria of the American College of Rheumatology
(ACR/SLICC) revised criteria of SLE [5]. Patients with
drug-induced SLE or pure cutaneous lupus were excluded.
Patients with diagnosis of SLE at the age of 50 or later
were classified as the "late onset" lupus,
all patients aged younger than 50 years at SLE diagnosis,
constituted the ''early-onset'' SLE group. All patients
underwent a physical examination. In addition to routine
laboratory investigations including tests were conducted
for rheumatoid factor, antinuclear antibody (indirect
immunofluorescence method with HEp-2 cells as substrate),
complements 3 and 4, anticardiolipin antibodies, and
lupus anticoagulant. Other several examinations such
as an echogram and computerized tomography were performed,
if needed. The following items were recorded: age at
the onset of the disease, sex, date of diagnosis, clinical
and laboratory manifestations and the status at the
last visit. Organ involvement not related to SLE was
excluded. The SLE disease activity index (SLEDAI) was
used to measure the severity of disease at the time
of diagnosis [6].
Statistical analysis: The
data were analyzed statistically using SPSS software
for Windows (version 18) to compare the epidemiological,
clinical and laboratory parameters in different groups.
A Fisher exact test was performed for qualitative variables,
and a Student t-test for comparison of quantitative
variables. Statistical significance was defined as p<0.05.
Results
Baseline characteristics and clinical manifestations
at the time of onset of SLE.
From the total of 83 (75 female and 8 male) patients
included in this study, 12 patients were identified
as having late-onset SLE; mean age at SLE diagnosis
was 58.4 ± 5.8 years (range, 51-72 yr.). The
early onset control group n= 71 (mean age 31.8 ±
10 years, range 16-49). There was no significant difference
in gender distribution among the groups (Table 1).
Table 1 : Demographic characteristics of SLE in late
and early onset patients
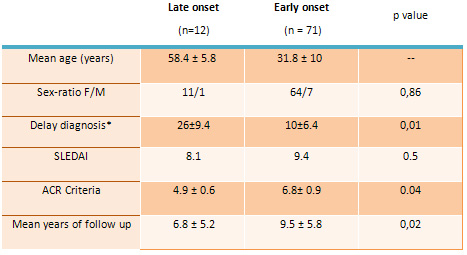
*delay between SLE onset and diagnosis, F/M: female/male
The duration between disease onset and diagnosis was
26 months (range, 11-48) for the old patients, compared
with 10 months (range, 3-24) in the younger SLE group.
This difference was statistically significant (p=0.01).
Mean number of revised ACR criteria was smaller for
the older population compared to the younger one (4.9
± 0.6 vs 6.8± 0.9; p = 0.04),
Clinical manifestations at diagnosis
in both groups are summarized in Table 2.
Table 2 : Clinical manifestations at the diagnosis
of SLE in late and early onset patients
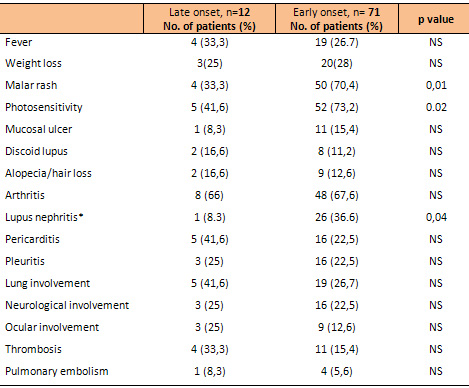 *Proliferative
glomerulonephritis (WHO class II, III or IV) *Proliferative
glomerulonephritis (WHO class II, III or IV)
In the Late onset group lupus showed a significantly
less frequency of malar rash (33.3% vs 70.4 %; p=0.01),
photosensitivity (41.6% vs 73.2 %; p=0.02) and lupus
nephritis (8.3 % vs 36.6 %; p= 0.04) compared with the
early-onset group.
Laboratory
findings
Regarding laboratory findings at the time of onset of
SLE (Table 3), the late-onset group was characterized
by a lower frequency of leukopenia (25% vs 55 %; p=0.05).
The other hematological disorders (lymphopenia, hemolysis
anemia and thrombocytemia) were slightly less frequent
compared to the early-onset group, but differences were
not significant. Whereas rheumatoid factor positivity
occurred more frequently in the elderly. No significant
differences were found for anti-dsDNA, anti-Sm, and
antiphospholipid antibodies.
Table 3: Laboratory findings of SLE in late and early
onset patients
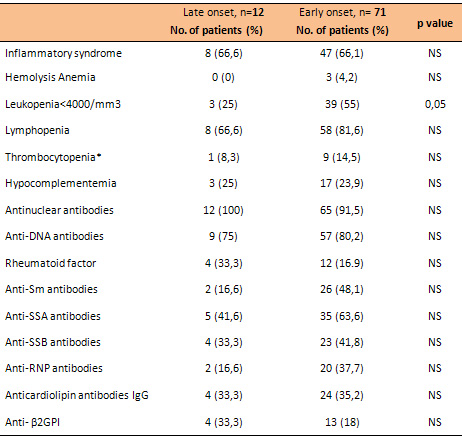
*< 100000/ mm3,
Table 4 summarizes the main
concomitant auto-immune diseases associated to SLE.
The late onset of lupus was associated with increased
incidence of rheumatoid arthritis (33.3 % vs 7%; p=0.007).
APS and Sjogren syndrome were slightly more frequent
compared to the early-onset group.
Table 4 : Autoimmune diseases associated with SLE
in late and early onset patients
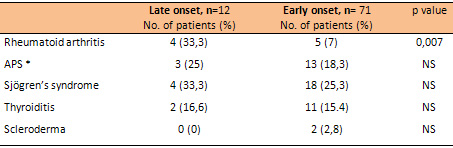
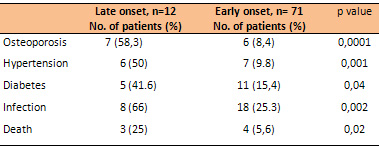
Table 5 : Complications that occurred during follow
up in late and early onset patients
Therapy
and course
High-dose corticosteroids >1mg/kg/day (42.2
% vs. 18 %; p = 0.05) and hydroxychloroquine (92.9 %
vs 75 %; p = 0.007) were less frequently used in the
late-onset group.
The late onset of lupus was associated with increased
incidence of hypertension, diabetes, osteoporosis and
infections. At the time of analysis, 3 patients had
died in the late onset group of which 3 were infections,
and only 4 patients in the early onset group (p=0.02).
None of our late-onset SLE patients died of SLE flare.
Discussion
SLE has been considered to be a disease of young women.
Late onset SLE is less frequent. From the total of 83
SLE patients in this study, twelve late-onset SLE patients,
that is, diagnosed at or over the age of 50 years, represented
14.4% of all patients with SLE followed in our department.
Different incidences have been reported (3.6-20 %).
This may be related to the lack of strict definition
of late-onset SLE. The cutoff age used most frequently
is 50 years at disease or diagnosis [1,2, 7,8]. The
female-to male ratio declines but not significantly
with age in our study. In most of the literature data,
the female to male ratio was significantly lower in
the late onset group [7]. This probably reflects the
relationship between SLE and estrogen status [8-9].
Oestrogens have multiple immunomodulatory effects. There
was a significant delay in the diagnosis of SLE in older
ages. This longer interval may be related to the atypical
presentation of SLE in the older population. The disease
seems to be mild with lower SLE criteria compared to
the younger group. Similar results have been reported
by many other studies [8, 10].
Age at disease onset of SLE influenced the clinical
manifestations and serological laboratory findings.
The result of our study shows that older population
less often presented with malar rash, photosensitivity,
and nephropathy. Most of the literature data segregating
early and late onset group, show lower rates of nephritis,
malar rash, photosensitivity, arthritis and less visceral
involvement with more benign course in the older group,
whereas, pulmonary involvement and serositis are more
frequently observed [8, 10, 11, 12] . The results of
our study support the concept that late onset lupus
is less active in the older population [10- 16].
Regarding laboratory results,
Leukopenia was significantly more frequent in the early
onset group, whereas rheumatoid factor positivity was
more frequent (33.3 % vs. 16.9 %) in old compared with
young SLE patients, but the difference was not statistically
significant, although no difference was found for other
autoantibody frequencies (anti DNA, anti-Sm, anti-RNP,
SSA and anti-SSB antibodies). Das Chagas Medeiros MM
et al reported leuko/lymphopenia most frequently in
late onset lupus but Appenzeller et al. reported hemolytic
anemia and thrombocytopenia most frequently in late-onset
SLE. Lalani et al. found no difference between the groups
when comparing lymphopenia and thrombocytopenia [15,
17-18].
It should be noted that, it
is difficult to be conclusive with regard to autoantibody
findings, since they were not measured in the same laboratory.
For the rheumatoid factor, most publications agree in
indicating its higher frequency in patients with late-onset
SLE [7, 19, 20], However, it should be noted that this
is probably related to the prevalence of increased rheumatoid
factor positivity in the elderly population. The literature
is rather inconsistent with regard to the predominant
serological profile in early and late onset SLE. Our
study found no differences in the autoantibody profile
in the two groups. Boddaert J et al reported a higher
frequency of anti-RNP, anti-Sm and rheumatoid factor
with a lower frequency of hypocomplementemia in late-onset
SLE [7]. Appenzeller et al. reported a lower frequency
of anti-Ro in late-onset SLE, while Alonso MD et al
reported a lower frequency of positive anti-DNA [17,21].
Lower positive rates of autoantibodies and less frequent
hypocomplementaemia may indicate milder disease activity
in older population [3, 21]. Some series reported a
higher prevalence of positive anti-Ro/SSA and/or anti-La/SSB
antibodies [22-25]. In the present investigation, there
were no differences in the frequency of Sjogren's syndrome
among the group of late-onset SLE and the group of early-onset
SLE patients, but rheumatoid arthritis was more associated
to late onset SLE. Another cohort reported a higher
frequency of Sjogren's syndrome in their populations
with late-onset SLE [19, 27]; Boddaert J et al in their
pooled data analysis, reported that Sjögren's syndrome
was more frequently present in the late-onset than in
the early-onset SLE, although no difference was found
for anti-SSA and anti-SSB antibodies [7]. The lack of
a parallel increase in Sjogren's autoantibodies in these
patients may reflect the aging process, in fact the
sicca symptoms may be related to other more frequent
causes of mucosal dryness in the elderly population,
such as senile salivary gland atrophy, polypharmacy
and chronic debilitating diseases [27, 28].
Hypertension, diabetes and osteoporosis
were significantly more increased in the late onset
group than in the early onset group in our cohort. The
greater frequency of age related comorbidities in older
population is due to aging and longer exposure to classical
risk factors and use of corticosteroids [7,29]. In our
cohort, the mortality rate was greater in the late onset
group; infection caused death in 3 of 4 patients. The
higher mortality in older patients contrasting with
the lower severity of the disease, may be related to
the fact that lupus at that age may be complicated by
co-morbidities and increased risk of toxicities from
immunosuppressive drugs usually used.
In our study glucocorticoids, hydroxychloroquine use
was less frequent in the late-onset group compared to
the early-onset group.
Our study has some limitations.
First the validity is limited by the retrospective design;
the collection of the data may have underestimated the
frequency of some clinical manifestations. Second, the
size of our sample is quite small; it is representative
only for the population of the center of Tunisia. We
also acknowledge that there may have been changes to
medications or co-morbidities outside our hospital.
In summary, this study
in the Tunisian SLE population showed that age affects
the expression of SLE. Late onset SLE has a milder disease
compared with early onset group, characterized by less
renal, photosensitivity and malar rash. However, the
mortality rate of older population is higher. Co-morbidities
such as hypertension, diabetes and osteoporosis were
significantly more frequent in late-onset SLE.
References
1-Louzir B, Othmani S,
Ben Abdelhafidh N et al. Le lupus érythémateux
systémique en Tunisie.
Étude multicentrique nationale. Rev Med Interne
2003; 24: 768-774.
2-Maddison P.J. Systemic lupus erythematosus
in the elderly. J Rheumatol 1987; 14: 182-187.
3- Chen TL, Wong CH, Lee Ch et al. Systemic lupus erythematosus
in the elderly. Gerontology
2009 ; 3:108-113.
4-Dieval C, Ribevio O, Grenouillet MD et al. Lupus du
sujet âgé : étude descriptive clinic
biologique à partir de 44 cas. Rev Med Interne
2008 ; 29 : 294-336 .
5- Petri M, Orbai AM, Alarcon GS, Gordon C, Merrill
JT, Fortin PR, et al. Derivation and validation of the
Systemic Lupus International Collaborating Clinics classification
criteria for systemic lupus erythematosus. Arthritis
Rheum. 2012;64(8):2677-86.
6-Gladman D, Ginzler E, Goldsmith C, Fortin P, Liang
M, Urowitz M, et al. The development and initial validation
of the Systemic Lupus International Collaborating Clinics/American
College of Rheumatology damage index for systemic lupus
erythematosus. Arthritis Rheum. 1996;39(3):363-9.
7- Boddaert J, Huong DL, Amoura Z, Wechsler B, Godeau
P, Piette JC. Late-onset systemic lupus erythematosus:
a personal series of 47 patients and pooled analysis
of 714 cases in the literature. Medicine. 2004;83(6):348-59.
8-Sassi RH, Hendler JV, Piccoli GF, Gasparin AA, da
Silva Chakr RM1, Brenol JC et al. Age of onset influences
on clinical and laboratory profile of patients with
systemic lupus erythematosus. Clin Rheumatol. 2017 ;36(1):89-95.
9- Cartella S, Cavazzana I, Ceribelli A, Inverardi F,
Tincani A, Franceschini F. Evaluation of mortality,
disease activity, treatment, clinical and immunological
features of adult and late onset systemic Lupus erythematosus.
Autoimmunity. 2013;46(6):363-8.
10-Choi JH, Park DJ, Kang JH, Yim YR, Lee KE, Lee JW
et al. Comparison of clinical and serological differences
among juvenile-, adult-, and late-onset systemic lupus
erythematosus in Korean patients. Lupus 2015, 24 (12),
1342-49.
11-Medlin JL1, Hansen KE2, Fitz SR3, Bartels CM. A systematic
review and meta-analysis of cutaneous manifestations
in late- versus early-onset systemic lupus erythematosus.
Semin Arthritis Rheum. 2016 Jun;45(6):691-7.
12-Zulfiqar AA1, Courtel T1, Novella JL1, Pennaforte
JL1. Late-onset lupus in the elderly after 65 years:
retrospective study of 18 cases. Geriatr Psychol Neuropsychiatr
Vieil. 2015 ;13(2):157-68
13- Stefanidou S, Gerodimos C, Benos A, Galanopoulou
V, Chatziyannis I, Kanakoudi F, et al
Clinical expression and course in patients with late
onset systemic lupus erythematosus. Hippokratia. 2013
;17(2):153-6.
14- Sousa S, Gonçalves MJ, Inês LS, Eugénio
G, Jesus D, Fernandes S et al. Clinical features and
long-term outcomes of systemic lupus erythematosus:
comparative data of childhood, adult and late-onset
disease in a national register. Rheumatol Int. 2016
;36(7):955-60.
15- das Chagas Medeiros MM, Bezerra MC, Braga FN, da
Justa Feijão MR, Gois AC, Rebouças VC,
et al . Clinical and immunological aspects and outcome
of a Brazilian cohort of 414 patients with systemic
lupus erythematosus (SLE): comparison between childhood-onset,
adult-onset, and late-onset SLE. Lupus. 2016 ;25:355-63.
16-Catoggio LJ, Soriano ER, Imamura PM, Wojdyla D, Jacobelli
S, Massardo L, et al. Late-onset systemic lupus erythematosus
in Latin Americans: a distinct subgroup? Lupus 2015;24(8):788-95
17- Appenzeller S Pereira DA, Costallat LT. Greater
accrual damage in late-onset systemic lupus erythematosus:
A long-term follow-up study. Lupus 2008; 17: 1023-28
18- Lalani S, Pope J, de Leon F, Peschken C;. Members
of CaNIOS/1000 Faces of Lupus. Clinical features and
prognosis of late-onset systemic lupus erythematosus:
Results from the 1000 faces of lupus study. J Rheumatol
2010; 37: 38-44
19- Maddison P, Farewell V, Isenberg D, The rate and
pattern of organ damage in late onset systemic lupus
erythematosus. J Rheumatol 2002; 29: 913-17
20- Peñaranda-Parada E, Quintana G, Yunis JJ,
Mantilla R, Rojas W, Panqueva U et al
Clinical, serologic, and immunogenetic characterization
(HLA-DRB1) of late-onset lupus erythematosus in a Colombian
population.Lupus. 2015 ;24:1293-9
21-Alonso MD, Martinez-Vazquez F, de Teran TD, Miranda-Filloy
JA, Dierssen T, Blanco R et al. Late-onset systemic
lupus erythematosus in Northwestern Spain: differences
with early-onset systemic lupus erythematosus and literature
review. Lupus. 2012 Sep;21(10):1135-4821-
22- Mak SK, Lam EKM, Wong AKM. Clinical profile of patients
with late-onset SLE: not a benign subgroup. Lupus 1998;
7: 23-28.
23- Shaikh SK, Wang F. Late-onset systemic lupus erythematosus:
clinical and immunological characteristics. Med J Malaysia.
1995;50:25-31.
24- Ward MM, Studenski S. Age associated clinical manifestation
of SLE: a multivariate
regression analysis. J Rheumatol 1990;17:476-81.
25- Lazaro D. Elderly-onset systemic lupus erythematosus:
prevalence, clinical course and
treatment. Drugs Aging: 2007; 24(9):701-15.
26-Strickland RW, Tesar JT, Berne BH, Hobbs BR, Lewis
DM, Welton RC. The frequency of
sicca syndrome in an elderly female population. J Rheumatol
1987; 14: 766-71.
27- Padovan M, Govoni M, Castellino G, Rizzo N, Fotinidi
M, Trotta F. Late onset systemic
lupus erythematosus: No substantial differences using
different cut-off ages. Rheumatol
Int 2007; 27: 735-41
28- Achour A, Mankaï A, Thabet Y, Sakly W, Braham
F, Kechrid C, et al. Systemic lupus erythematosus in
the elderly. Rheumatol Int. 2012 ;32:1225-9.
29- Merola JF, Bermas B, Lu B, Karlson EW, Massarotti
E, Schur PH, Costenbader KH.
Clinical manifestations and survival among adults with
(SLE) according to age at diagnosis.
Lupus. 2014 ;23:778-84
30- Abu-Shakra M, Urowitz
MB, Gladman DD, Gough J. Mortality studies in systemic
lupus erythematosus. Results from a single center. I.
Causes of death. J Rheumatol. 1995;22:1259-64.
|

